R2A Testing for Dental Waterlines: Why StandardCheck™ Is the Gold Standard
Dental unit waterlines (DUWLs) are essential for patient care, but they can also be a hidden source of contamination. These narrow, low-flow tubes create ideal conditions for biofilm formation, which harbors bacteria and other microorganisms. Without proper maintenance and monitoring, water delivered during patient care can exceed safe microbial limits.
The CDC, ADA, and ADS (formerly OSAP) all recommend that water used in nonsurgical dental procedures meet the EPA drinking water standard of ≤500 CFU/mL of heterotrophic bacteria. Achieving this benchmark requires two things: routine treatment of waterlines and regular monitoring using validated test methods.
What ‘R2A’ Actually Means
R2A agar is short for Reasoner’s 2A agar. Agar is a gelatin like material that creates a stable surface to grow bacteria on. Reasoner’s 2A is a low-nutrient medium (essentially food) specifically designed to help identify bacteria in treated water systems. Unlike rich media that favor fast-growing organisms, R2A supports the growth of stressed or chlorine-tolerant bacteria, the same organisms that persist in dental waterlines. Instead of growing everything, it focuses on growing types of bacteria that commonly grow in water.
Here’s why this matters:
Dental waterlines use treated water (municipal or distilled), so surviving bacteria are often adapted to low-nutrient environments.
Biofilm organisms can be slow-growing, and R2A’s extended incubation period (typically 5–7 days) allows these colonies to develop.
EPA and APHA Standard Methods recognize R2A as the preferred medium for heterotrophic plate counts (HPC) in potable water systems.
In short, the R2A Agar provides a more accurate picture of contamination than quick-growth media, making it the method of choice for dental waterline testing.
What Sets StandardCheck™ Apart
Even though the R2A test method is a Standard Method, there are different ways to execute the test. Agenics Labs designed StandardCheck™ use the most advanced practices, ensuring high accuracy and dependability.
Here’s what sets it apart from other R2A tests in the industry:
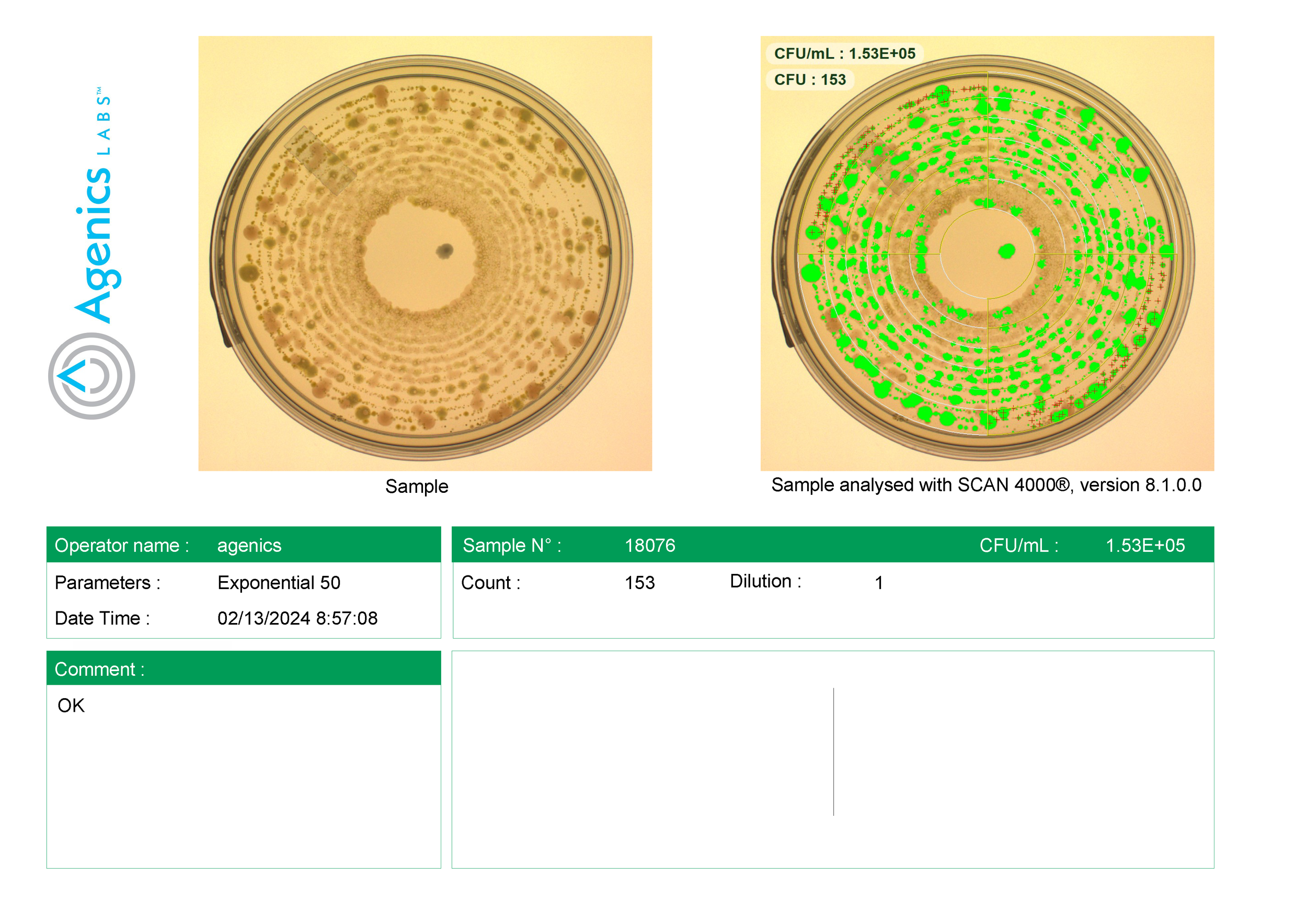
Photo Documentation (Agenics Exclusive): Only Agenics images plates so you can see the actual colonies behind the CFU count. (See photos in example report.)
Automated Digital Counting: Our state of the art digital colony counter eliminates human error due to manual colony counting.
Exponential Dilution Plating: Provides accurate enumeration of bacteria whether the sample is bacteria-free or contains over one million colonies and prevents “too numerous to count” results.
The Standardized Method requires a plated sample to contain less than 300 CFU for acceptable counting, which means dilutions must be performed to accurately report on any sample >300 CFU/mL.
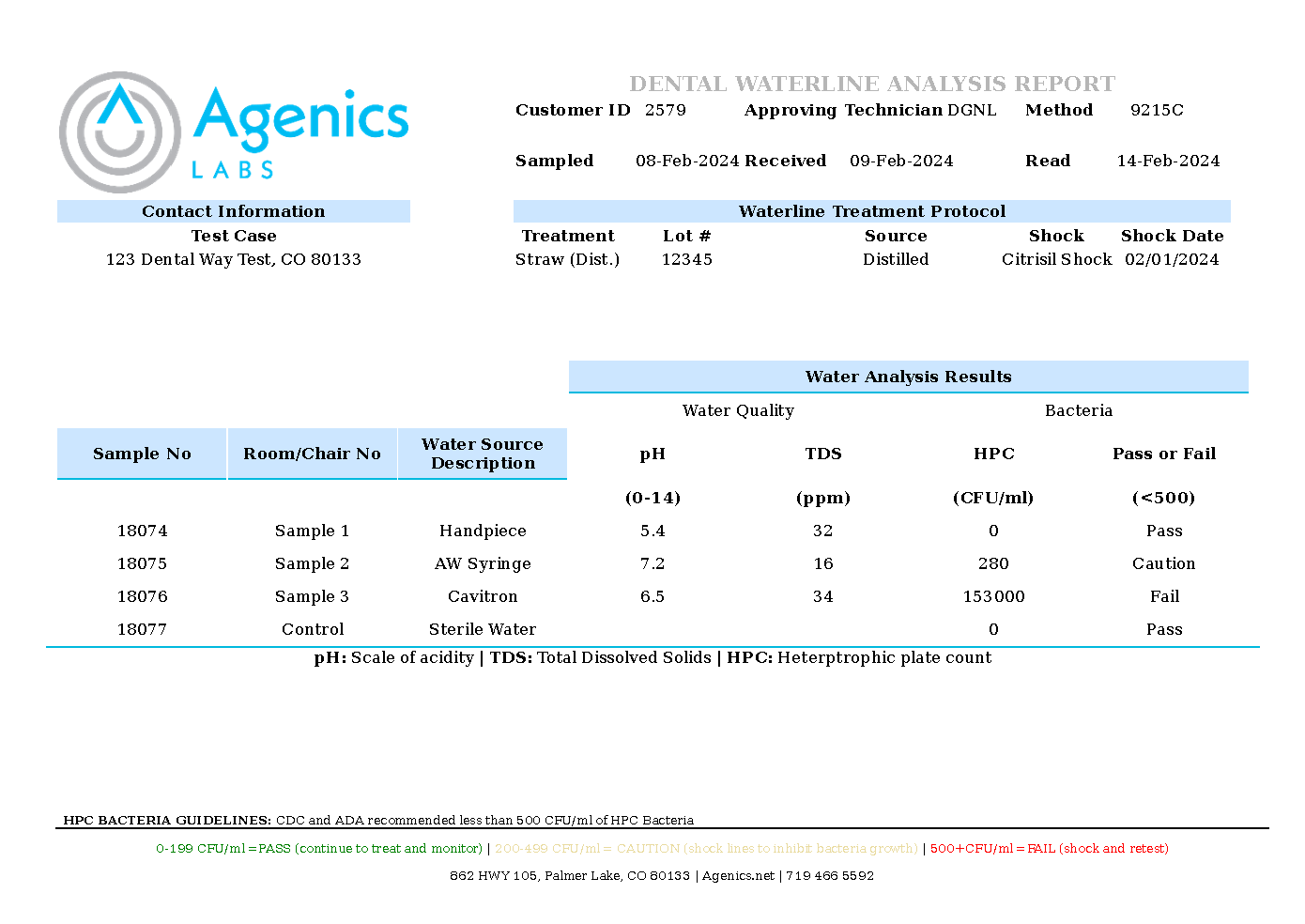
Expanded Water Quality Panel (Agenics Exclusive): Includes pH and TDS readings to help diagnose compatibility issues and rapidly troubleshoot failure root causes.
Digital Portal: Access reports and images anytime for internal review, audits, or staff training.
Convenient Logistics: StandardCheck™ kits include sterile vials, an insulated shipper, freezer packs, and an overnight return label to make sample collection seamless.
Example report showing pH,TDS,HPC count
Images of the R2a plates used in the report. (Exclusive to Agenics)
Regulatory and Professional Guidelines
Several organizations set expectations for dental water quality:
CDC: Water for nonsurgical procedures should meet ≤500 CFU/mL. Sterile water or saline must be used for surgical procedures.
ADA: Reinforces the CDC standard and emphasizes routine monitoring and documentation.
ADS (formerly OSAP): Advocates for validated test methods (such as R2A testing), written protocols for testing and remediation, and staff training.
EPA: Recognizes R2A agar for heterotrophic plate counts in treated water systems.
These guidelines share a common theme: treat, test, document, and remediate. Compliance isn’t just about checking a box, it’s about risk management and professional responsibility.
How the R2A Method Works (In Depth)
A water sample may be full of bacteria, yet look completely clean. The fundamental purpose of traditional culture based test methods, is to feed the existing bacteria, helping it multiply and grow into visible colonies.
Heterotrophic plate count (HPC) testing is a culture based enumeration method in which a known volume of water is distributed onto a low-nutrient agar surface (in this case, Reasoner’s 2A agar) and incubated for an extended period to promote the recovery and growth of slow-growing, stressed heterotrophic bacteria. This process results in discrete, countable colonies.
Methodology:
1. Homogenization and dilution strategy: The sample is mixed to distribute bacteria evenly. When higher counts are expected, the lab prepares serial dilutions, often 10 fold, so at least one plate falls within a countable range. This prevents confluent growth that can lead to “too numerous to count” results and protects the accuracy of the final CFU/mL.
2. Plating on R2A (HPC): A measured aliquot, commonly 0.1 mL for a spread plate, from the undiluted sample and or selected dilutions is applied to the R2A agar surface and spread for even distribution. Each viable cell, or a small cluster of cells, that can reproduce under these conditions can form a visible colony.
3. Incubation and recovery: Plates are incubated for an extended period, typically 5 to 7 days. The low nutrient formulation of R2A, combined with longer incubation, supports recovery of oligotrophic and disinfectant stressed heterotrophs that are commonly found in treated water systems and within dental unit waterlines. Faster methods and richer media can undercount these organisms when they do not resuscitate well or are outcompeted by faster growers.
4. Colony enumeration and CFU/mL calculation: After incubation, colonies are counted with validated imaging software. Results are reported as colony forming units per milliliter (CFU/mL) and calculated using the colony count, plated volume, and dilution factor. Viable but nonculturable organisms are not counted, and cell clumps can form a single colony, which can make CFU/mL lower than the true number of individual cells.
5. Quality controls and countability rules: The lab uses negative controls as sterility tests for quality control rather than blank media. Replicates or multiple dilutions may be plated to ensure that at least one plate is countable. Very low counts are more sensitive to random variation, and plates with confluent growth cannot be enumerated accurately. Incubation temperature, incubation duration, plating volume, and how mixed colony morphologies are counted can all affect the final CFU/mL. For trending over time, consistency in plating, incubation, analysis, and handling is essential.
How R2A (culture/HPC) differs from flow cytometry
R2A HPC reports the culturable portion of heterotrophic bacteria that can reproduce on low nutrient agar under defined incubation conditions. This is the basis for the familiar ≤500 CFU/mL dental waterline benchmark. Flow cytometry measures cells detected optically, often using fluorescent stains, and can include intact but nonculturable organisms and injured cells, depending on staining and gating. Because these methods measure different targets, results will not convert 1 to 1. RapidCheck by Agenics Labs is designed to minimize false Passes, which means it is common for these flow cytometry counts to be higher than the StandardCheck R2A results.
Practical interpretation: Use R2A CFU/mL (StandardCheck) to assess whether the water meets the established culture based benchmark and whether the system is supporting bacterial regrowth. Use flow cytometry (RapidCheck) to understand the total bacterial cell count, including organisms that may not grow on agar, and to help identify residual microbial load that culture based testing can miss.
Testing Implementation
When to Test
Routine monitoring is essential for compliance and patient safety. CDC and ADS recommend regular testing, but best practices for dental offices includes the following:
Quarterly testing for full documentation and compliance
Post-remediation testing after shock treatments
Testing after maintenance or plumbing\equipment changes
Increased frequency if you serve high-risk patients, experience persistent failures, or have variable source water quality
Check your state regulations here.
What do When Results Exceed 500 CFU/mL
Don’t panic! Testing allows you to proactively monitor and control your lines. Here’s what to do:
Review reports to see if you have any trends.
Shock waterlines per manufacturer instructions.
Retest to confirm remediation.
See our document ACT STAT on responding to water test failures.
Key Takeaways
R2A testing method is considered the gold standard for dental waterline testing.
≤500 CFU/mL is the benchmark for drinking water.
StandardCheck™ delivers accurate results, photos, and chemistry data.
Use validated testing and document thoroughly. Agenics Labs makes compliance easy with comprehensive testing solutions and expert support.
Agenics Labs as Your Compliance Partner
Beyond testing, Agenics Labs offers:
Educational resources on sampling and remediation.
Multiple test options (StandardCheck™, RapidCheck™, SimCheck™) to fit your workflow.
Expert consultation for persistent failures or protocol review.
Our goal is simple: help you maintain safe waterlines with confidence and efficiency.
Ready to Strengthen Your Water Quality Program?
Order StandardCheck™ today
-
CDC Guidelines
· Centers for Disease Control and Prevention. (n.d.). Best practices for dental unit water quality. https://www.cdc.gov/dental-infection-control/hcp/dental-ipc-faqs/best-practices-dental-unit-water-quality.html
Centers for Disease Control and Prevention. (n.d.). Dental unit water quality. https://www.cdc.gov/dental-infection-control/hcp/summary/dental-unit-water-quality.html
ADA Recommendations
· American Dental Association. (n.d.). Dental unit waterlines. https://www.ada.org/resources/ada-library/oral-health-topics/dental-unit-waterlines
ADS (formerly OSAP) Guidance
· Association for Dental Safety. (n.d.). Dental unit waterlines (DUWL): OSAP recommendations to clinicians. https://www.myads.org/topics-duwl-osap-recommendations-to-clinicians
Association for Dental Safety. (n.d.). Home. https://www.myads.org/home-